 
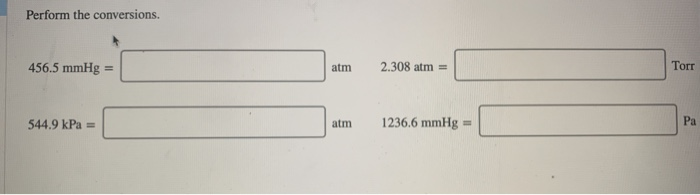
It is released and rises to an altitude of 8. A helium-filled weather balloon has a volume of 616 L at 22.9☌ and 757 mmHg. atm: Non-SI (International) 1 atm 101,325 Pa: pounds per square inch: psi or lbf/in 2: Non-SI (International) 1 psi 6,894. For case "A", the level of the liquid in the left side of the tube is lower than the level at the right side. P1 752 mmHg / 760 0.989 atm P2 522 mmHg / 760 0.687 atm Substitute known values into the combined gas law and solve for V2: (0.989 atm × 811 L) / 293.05 K (0.687 atm × V2) / 266.05 K. The liquid is placed in the "U" shaped tube connected to the big circle containing the gas. P A = atmospheric pressure, P Hg = pressure of mercury column P gas = pressure of confined gas. (a) Measuring a pressure greater than atmospheric (b) measuring a pressure less than atmospheric. A: Given: Temperature 35.0 oC 308 K Pressure 757 mmHg 0. Looking for a conversion Select a conversion. When placed in the same container, the total pressure is 1 atm, with each gas contributing its partial pressure. Alone, Hydrogen has a pressure of 0.4 atm, while Nitrogen has a pressure of 0.6 atm. Non-SI (International) 1 psi 6,894.757 Pa: millimeters of mercury: mmHg: Non-SI (International) 1 mmHg 133.3224 Pa: bar: bar: Non-SI (International) 1 bar 100,000 Pa: torr: Torr: Non-SI (International) 1 Torr 133.3224 Pa: Find a Conversion. 1: Above, the figure shows how Daltons law of partial pressure works. Type in unit symbols, abbreviations, or full names for units of length, area, mass, pressure, and other types.\) Use of a mercury manometer. 1 atm 101,325 Pa: pounds per square inch. You can find metric conversion tables for SI units, as well as English units, currency, and other data. The unit is named after Evangelista Torricelli, Italian physicist and mathematician, for his discovery of the principle of the barometer in 1643.Ĭ provides an online conversion calculator for all types of measurement units. It is the atmospheric pressure that supports a column of mercury 1 millimetre high. The torr (symbol: Torr) or millimetre of mercury (mmHg) is a non-SI unit of pressure. 
It is approximately equal to Earth's atmospheric pressure at sea level. Atmospheres : The standard atmospheric (symbol: atm) is a unit of pressure which is a non-SI units. 
It is widely used, and its value is approximately equal to a pressure of 1 torr. mmHg ( 0 c ) kPa 0.133 322 mbar kPa 0.1 stress kip / in2 MPa 6.894 757 ( formerly ksi ). Millimeter Mercury (0☌) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. It is sometimes used as a reference pressure or standard pressure. atm ( std ) kPa 101.325 bar kPa 100.00 lbf / in2 kPa 6.894 757. The standard atmosphere (symbol: atm) is a unit of pressure defined as 101325 Pa (1.01325 bar). Next, lets look at an example showing the work and calculations that are involved in converting from millimeters of mercury to torrs (mmHg to Torr).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
